Time to Retire Your Pharmacy Isolator?
For years, hospital pharmacies have relied on pharmacy isolators, including cumbersome and complex glove boxes, to compound sterile products. However, effective November 1, 2023, these isolators will lose practical benefits in areas of Beyond Use Dating (BUD) while gaining more stringent room placement requirements, placing them on par with other primary engineering controls (PECs) such as laminar airflow workstations (LAFWs) and Class II Biosafety Cabinets (BSCs). With major changes on the horizon, now is the time for your pharmacy to examine these new regulations and other challenges associated with pharmacy isolators. By proactively understanding these changes, you can ensure your pharmacy’s compliance and continue to provide safe and efficient compounding services.
What are Pharmacy Isolators?
Pharmacy isolators, also referred to as restricted-access barrier systems, or RABS for short per the new USP <797>, are enclosed PECs designed for compounding sterile products (CSPs). RABS come in two formats: Compounding Aseptic Isolators (CAIs) and Compounding Aseptic Containment Isolators (CACIs). CAIs are positive pressure devices for compounding sterile non-hazardous CSPs. CACIs are negative pressure devices for compounding of sterile hazardous drugs, including chemotherapy preparations, while minimizing the risk to personnel.
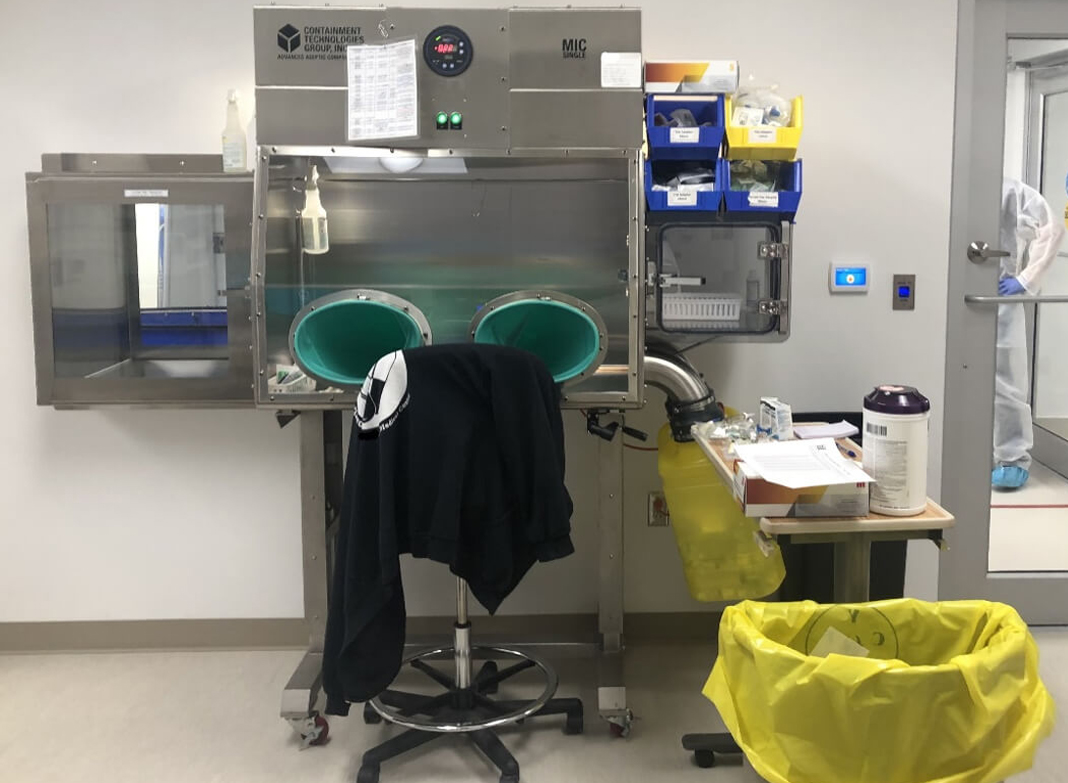
A CAI pharmacy isolator in an unclassified area of an inpatient pharmacy.
Both CAIs and CACIs offer a direct compounding area with HEPA filtered ISO 5 unidirectional (laminar) airflow. Materials are transferred in and out of these devices through an ISO 5 HEPA filtered transfer chamber. To access the primary working area, CAIs and CACIs are equipped with gloves located at the front of the isolator. These gloves enable pharmacy technicians to perform their compounding tasks while maintaining the necessary barrier between the operator and the sterile environment within the isolator.
New USP <797> BUD Restrictions
Although CAIs and CACIs will still be accepted as primary engineering controls (PECs) for sterile compounding in the new USP <797> update, a new change involves a shortening of the BUD for CAIs and CACIs placed outside of cleanrooms. CAIs and CACIs not placed inside a cleanroom suite with an ISO 7 buffer room and ISO 8 anteroom will only be granted a 12-hour BUD (24-hour BUD if refrigerated). Pharmacies compounding in this manner must classify these products as Category 1 CSPs.
CSPs that are made in isolators within a cleanroom suite with an ISO 7 buffer room and ISO 8 anteroom are granted a full BUD and Category 2 or 3 classification.
|
Isolator BUD Changes |
Allowable BUD |
|
|
Isolator Location |
Current <797> |
New <797> |
|
Unclassified Area |
Full BUD |
Category 1 CSP 12 Hours |
|
Cleanroom Suite (ISO 7 Buffer Room) |
Full BUD |
Category 2 or 3 CSP Full BUD |
In short - pharmacies that have previously relied on extended BUDs from CAIs and CACIs placed in the general unclassified will lose this benefit without a full cleanroom suite. LAFWs and BSCs now offer equal BUDs to CAIs and CACIs placed into equivalent areas.
New USP <800> Placement Requirements
CACI isolators that are used for compounding of hazardous sterile drugs must also comply with the placement requirements of USP <800>. For Category 1 CSPs, CACIs must be placed into a Containment-Segregated Compounding Area (C-SCA). C-SCAs are dedicated rooms with 0.01-0.03” w.g. negative pressure relative to adjacent areas, vented to the exterior, with a minimum of 12 Air Changes Per Hour (ACPH). Category 2 and 3 CSPs must be placed into a full cleanroom suite with a negative pressure ISO 7 buffer room and an ISO 8 anteroom.
|
USP <800> CACI Placement |
Old Requirement |
New USP <800> Requirement |
|
Room Type |
Unspecified |
C-SCA (Category 1 CSPs) |
|
Room Externally Vented |
Not Required |
Required |
|
Negative Pressure Room |
Not Required |
Required (0.01-0.03" w.g.)* |
|
Air Changes Per Hour (Minimum) |
Not Required |
12 (C-SCA) |
|
CACI Externally Vented |
Preferred |
Required |
*Relative to adjacent spaces
While the USP <800> placement requirement for CACIs also extends to Class II BSCs, CACIs require the same HD garbing, making them ergonomically unfavorable when working through the gauntlets and gloves that allow access to the direct compounding area. Class II BSCs are a logical improvement over CACIs because of their open sash and unrestricted working area.
Does My Isolator Have Unidirectional Airflow?
USP <797> requires that isolators provide unidirectional airflow in all pharmacy isolators. Unidirectional airflow as defined by CETA CAG-002 is described as, “…an airflow moving in a single direction in a robust and uniform manner and at sufficient speed to reproducibly sweep particles away from the critical processing or testing area.” Air that is not unidirectional is called turbulent airflow.
While there are many types of pharmacy isolators with HEPA filtered ISO 5 airflow, the airflow in some of these devices may be turbulent flow - not laminar/unidirectional. Turbulent airflow in the direct compounding area drastically removes the ability to compound in ISO 5 first air and puts patients at risk. Replacing a turbulent flow isolator with an ISO 5 unidirectional air PEC such as a LAFW or BSC improves patient safety.
Certification Difficulties
To ensure your pharmacy isolator is performing in a manner that ensures drug sterility and patient safety, isolators should undergo extra tests specific to isolators during each 6-month recertification process to comply with CETA CAG-002. These tests include successful completion of ingress and egress tests, plus a recovery test for the main chamber. For CACIs, a particle containment integrity test is also required.
These processes can leave your isolator out of commission for up to half a day and leave your pharmacy out an extra few hundred dollars on every re-certification compared to the requirements for certification of a LAFW or BSC
Other Pharmacy Isolator Challenges
Ergonomics: Any pharmacy technician that is compounding sterile products must take extreme care to ensure the integrity of their sterile products. A major factor in reducing errors is user comfort. Technicians working in CAIs and CACIs must handle all materials through fixed gloves in the front of the isolator. For users of medication workflow management system (e.g. Baxter DoseEdge, BD PharmacyKeeper, Omnicell IVX, etc.), operation may be heavily restricted through gloves with limited reach.
With CAIs and CACIs losing the benefit of longer BUDs without a cleanroom suite, pharmacies can utilize LAFWs and Class II BSCs in place of closed pharmacy isolator systems to give users more comfort and less chance for error.
Cleanability: Technicians using isolators are working through restrictive gloves that limit reach to all corners of the isolator, especially for users with shorter arms. Even with the use of a cleanroom mop, users may have difficulty cleaning every nook and cranny within an isolator. Compared to the open, accessible interior of a LAFW or BSC, pharmacy isolators are more difficult to clean.
Higher Lifetime Costs: Not only do CAIs and CACIs cost more to purchase than their LAFW and BSC counterparts, but they also require ongoing part maintenance that add additional operating costs over time. Plus, with higher certification costs, regular replacement of gauntlet arms, internal gloves and upkeep (especially for older isolators), these pieces of equipment can rapidly impact your pharmacy budget over time.
Operational Restrictions: Every opening of the transfer chamber brings contamination into the pharmacy isolator. Since pharmacy isolators are designed to operate with the transfer chamber door shut and the main window closed, they must undergo a recovery test that identifies the total time required for ISO 5 conditions to return within the isolator. Once validated, each technician must adhere to the recovery wait period. BSCs and LAFWs purge their work area rapidly, making this wait time a non-factor.
What Do I Need to do Next?
Each pharmacy has unique needs that impact PEC selection. While isolators are becoming less common, be sure to complete a full evaluation of your pharmacy hoods before moving forward with a change. Labconco has resources designed to get your pharmacy on track with PECs:
With the USP <797> and <800> deadline of November 1, 2023 fast approaching, the time to evaluate is now.
| chevron_left | Optimizing Freeze Drying for DMSO Solutions | Articles | Lyophilization, concentration or evaporation, which is best for my samples? | chevron_right |






